BStabG: Der neue dynamische Herstellerabschlag – eine sinnvolle Lösung für die Kontrolle der Arzneimittelausgaben?
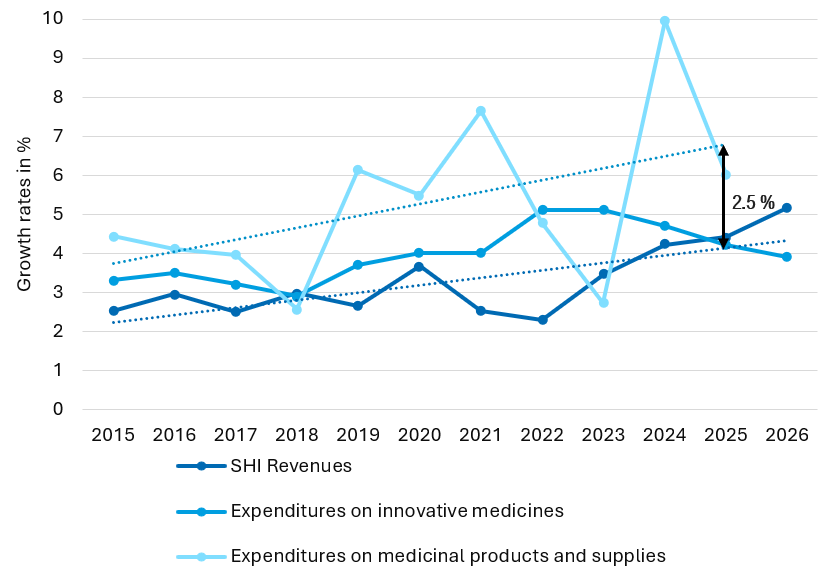
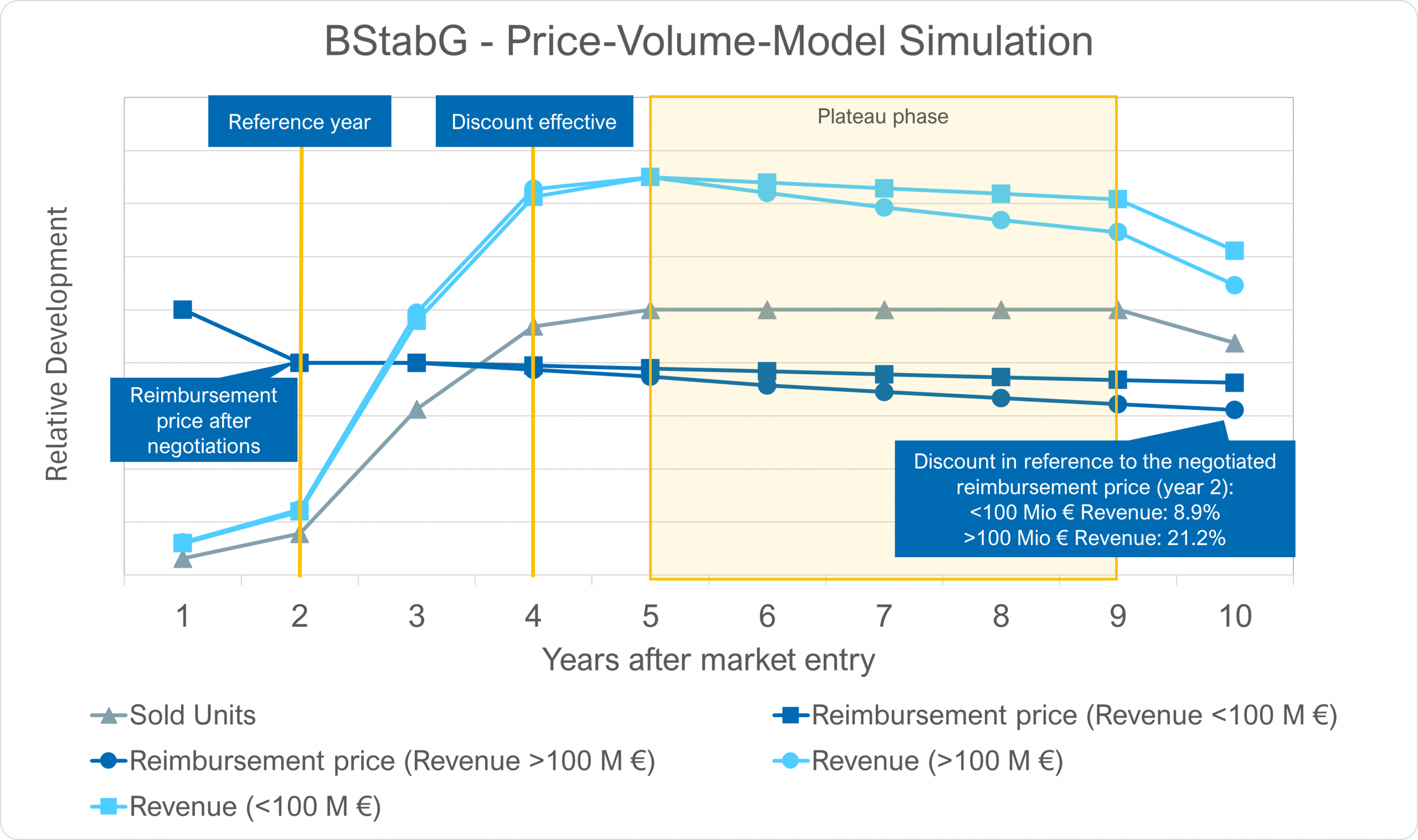
Der dynamische Herstellerrabatt ist ein ordnungspolitischer Fehltritt Die geplante Einführung eines dynamischen Herstellerabschlags für patentgeschützte Arzneimittel (§ 130a Abs. 1b SGBV) im Rahmen des BStabG wirft erhebliche ordnungspolitische Fragen auf – insbesondere im Kontext der bestehenden Sy…